We described our approach to sampling in our previous paper6. Briefly, we purchased thirty samples of rakia from small-scale and sustenance producers. None of the rakia samples had the tax stamp that appears on officially recorded products. We then transferred each sample into glass bottles with identification numbers to avoid any potential mismatches and stored them at 4 °C until we could conduct gas chromatographic mass spectrometric (GC/MS) and inductively coupled plasma optical emission spectrometric (ICP-OES) analyses. We recorded where each sample was purchased, the raw materials reported to be used in production, ethanol content as reported by the producers, price per litre, and the date of sampling.
Gas chromatographic mass spectrometric analysisMaterials
Acetonitrile and ethanol were supplied by Merck (Darmstadt, Germany) and used as internal and qualitative standards for the analysis of rakia samples, respectively. All reagents employed were of high-performance liquid chromatography grade.
Determination of ethanol concentration in rakia samples
We have described in detail how we determined ethanol concentration in our previous article6. Briefly, we analysed the rakia samples using a Hewlett-Packard (HP) GC/MS system (Palo Alto, CA, USA) consisting of a HP 6890 GC, a HP 5973 mass selective detector (MSD) and an Agilent 7683 automatic liquid sampler (Agilent Technologies, Palo Alto, CA, USA). A Hewlett-Packard Free Fatty Acid Phase (length: 50 m, internal diameter: 0.2 mm, film thickness: 0.33 μm) cross-linked capillary column (Hewlett-Packard, Palo Alto, CA, USA) was used for the separation of ethanol using helium as carrier gas at 3.0 bar constant pressure.
Inductively coupled plasma optical emission spectrometric analysisMaterials
Nitric acid and mono-element spectroscopic standards were obtained from ESLab (Debrecen, Hungary), while the multi-element spectroscopic standard solution and hydrogen peroxide were purchased from Merck (Darmstadt, Germany). Ultrapure water was prepared using a Synergy UV water purification system (Merck Millipore, Darmstadt, Germany). All reagents used were of spectroscopic or reagent grade.
Determination of element levels in rakia samples
The concentrations of 24 elements were determined using an Agilent ICP-OES system (model 5100 VDV, Agilent Technologies, Santa Clara, USA) as previously described in detail6. We used an autosampler (Agilent SPS4), a Meinhard nebuliser and a double-pass spray chamber to introduce the samples. Argon gas was used for plasma supply. Standard arsenic (As) and tin (Sn) solutions were prepared from mono-element spectroscopic standards with a concentration of 1000 mg/litre. In addition, multi-element standards containing all other elements at a concentration of 1000 mg/litre were used to prepare calibration solutions for silver (Ag), aluminium (Al), boron (B), bismuth (Bi), calcium (Ca), Cd, Co, Cr, Cu, iron (Fe), gallium (Ga), indium (In), potassium (K), lithium (Li), magnesium (Mg), manganese (Mn), sodium (Na), Ni, Pb, strontium (Sr), thallium (Tl), and zinc (Zn). A five-point calibration curve was used for the quantitative analysis. Three parallel measurements were carried out for each sample. To validate the ICP-OES method, blank samples were used to check the purity of water and glassware used. The influence of ethanol concentration on the detectability of heavy metals was determined by preparing solutions containing ethanol (Merck, Darmstadt, Germany) at concentrations of 0.0, 5.0, 10.0, 25.0, and 40.0% spiked with the multi-element standards. The final concentration of the elements was 0.1 mg/L. The accuracy of the measurements was above 95%. The operating conditions of the ICP-OES are given in Supplement 1.
Comparison of metal concentrations in unrecorded rakia with threshold values
The metal concentrations measured in the rakia samples were compared with the limits proposed by the AMPHORA project18. The levels of metals were expressed in milligrams per litre of pure alcohol (p.a.) to allow comparison with the results of previous research.
Statistical analysis of ethanol levels in rakia samples
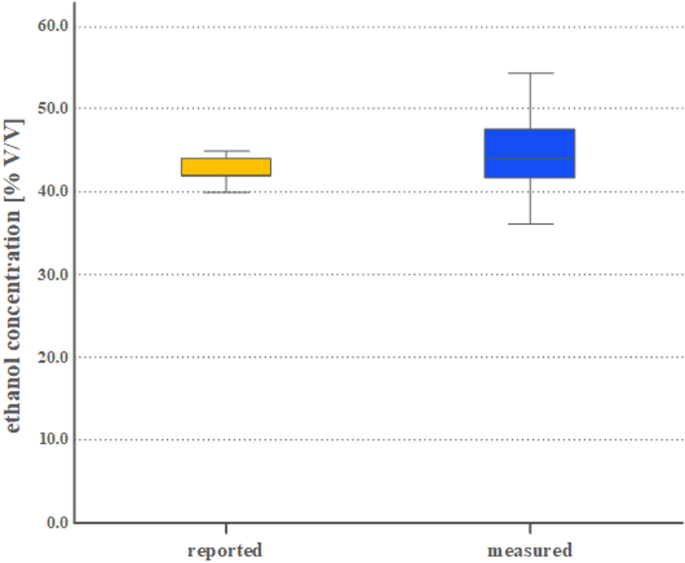
The ethanol levels reported by the providers of rakia samples (reported) were compared to those determined in the GC/MS analyses (measured). The distribution of the data was evaluated for normality using the Shapiro–Wilk test, which indicated that the data were not normally distributed. Therefore, we used the Wilcoxon signed-rank test to evaluate the mean differences between the reported and measured ethanol concentrations. We conducted the statistical analysis using IBM’s SPSS software version 25.0 (IBM Inc., Armonk, New York, USA). We considered a p-value of less than 0.05 to be statistically significant. The median ethanol concentration, along with its interquartile range (IQR) and 1.5 times the IQR (shown as whiskers), are illustrated in Fig. 1.
Estimation of health risks
The toxicological thresholds proposed by the AMPHORA project are based on extrapolation of drinking water quality guidelines and standards of the WHO and European Union (EU), respectively18. Assuming an average daily per capita consumption of alcohol ten times lower than that of drinking water, the AMPHORA limits were calculated by multiplying the WHO and EU guidelines by 10. Drinking water standards established by the WHO and EU are based on a number of toxicological and epidemiological considerations and indicate the concentrations of chemical contaminants, including heavy metals, in drinking water that do not pose a significant health risk to consumers, even in the case of lifetime exposure19. Therefore, it is unlikely that the consumption of unrecorded rakia containing concentrations of heavy metals below the AMPHORA limits can result in a health risk. Consequently, to determine the health risk associated with consuming unrecorded rakia, we estimated the daily intake of those metals that exceeded the AMPHORA threshold value in at least one of the samples collected. Accordingly, Cu, Fe, Ni and Pb were selected. In those instances where their concentration was below the limit of detection (LOD), the LOD values of the selected metals (see Table 1) were divided by two and the entries indicated in the database as “< LOD” were replaced with the result of the calculation. Next, the median and the 99th percentile values of the concentration of the selected metals (see Table 2) were calculated. Then these data were used to estimate the target hazard quotient (THQ) values if a reference dose had previously been reported for the selected metals5. As a result, the median and the 99th percentile values of the concentration of Cu, Fe and Ni were applied in the following formula:
$$EDI = \frac{MDI \times MCS}{{BW}}$$
where, the EDI is the estimated daily intake, the MDI is the mass of the daily ethanol intake in g/day, MCS is the median and the 99th percentile values of the concentration of Cu, Fe and Ni in mg/g of p.a. and BW is the average body weight of 73.9 kg for both sexes5. The health risk estimation considered two scenarios: “average drinkers” and “heavy drinkers”. Although data on the volume of unrecorded alcohol intake in the RK are not available, we assumed that it would be similar to the levels reported for both sexes aged 15 years and above in neighbouring Balkan countries, such as Albania (0.7 L of p.a./capita/year), Montenegro (0.7 L of p.a./capita/year), North Macedonia (0.8 L of p.a./capita/year), and Serbia (0.8 L of p.a./capita/year)4. Averaging these volumes, the intake of ethanol from homemade rakia in the RK was estimated to be 0.75 L of p.a./capita/year, assuming that all unrecorded alcohol was consumed in that form. Subsequently, the weight of unrecorded alcohol consumed annually was calculated by multiplying 0.75 L by the density of ethanol (0.789 g/cm3), resulting in a quantity of 591.8 g/year. This is equivalent to the consumption of 1.62 g of ethanol per day. This value was used in the scenario termed “average drinkers”. The second scenario, termed “heavy drinkers”, was defined as the consumption of 60 g of ethanol per day in unrecorded rakia4. Subsequently, THQ values were calculated5. The THQ is a method used to estimate the non-carcinogenic risk related to chronic exposure to chemicals such as metals5. It is calculated as the ratio of the oral dose of a metal to its reference level5. A ratio exceeding 1.0 is interpreted as an elevated health risk, while values less than 1.0 are considered negligible5. To calculate the THQ values, the following factors were taken into account: the EDI, the exposure frequency (EF, 365 days/year), the exposure duration (ED, years), the average exposure time (AET, 365 days/year × ED) and the oral reference dose of the particular metals (RfD, mg/kg/day, see Table 1)5,20. The ED was defined as the number of years that an average Kosovar person can be expected to live at the age of 15 (65.4 years for both sexes)21. The following equation was used for the health risk estimation:
$$THQ = \frac{MCS \times MDI \times EF \times ED}{{RfD \times BW \times AET}}$$
Since a RfD value for Pb has not been reported, the margin of exposure (MOE) approach was applied to estimate the health risk associated with drinking Kosovar rakia containing this heavy metal22. As recommended by the European Food Safety Authority (EFSA), this is the preferred method for assessing the risk to human health from the presence of Pb in food16. The MOE can be defined as the ratio of the lower bound of the benchmark dose confidence interval (BMDL) of a substance to the estimated intake of the same substance16,22. As the MOE value highly depends on the health endpoints for which the BMDL was determined, we considered those outcomes that were found to be the most critical in adults, including cardiovascular effects and nephrotoxicity using BMDL values of 0.0015 (BMDL01) and 0.00063 (BMDL10) mg/body weight kg/day, respectively16. MOE values less than 100 are considered as a public health concern for substances not classified as genotoxic or carcinogenic, including Pb22. The MOE values were calculated as follows:
$$MOE=\frac{BMDLx}{EDI}$$
where the BMDLx is the BMDL value reported for a specific health endpoint.