The RESILIENT dataset10 was collected using an open-source software platform9 that integrates data from wearable and remote in-home devices for continuous health monitoring. Data collection involved ethically approved procedures with informed consent from participants, and the final dataset was fully de-identified to ensure privacy and confidentiality.
RESILIENT platform
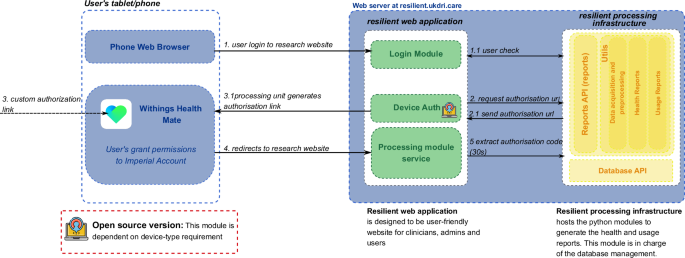
The RESILIENT platform9 is an open-source digital solution designed to integrate data from remote healthcare monitoring devices, providing comprehensive clinical visualisations and generating insightful reports for both users and healthcare professionals. The system follows a structured architecture to acquire data from Withings wearables11, leveraging the official Withings API, which provides access to multiple device-specific measurements.
The platform specifically integrates two Withings devices (Table 1): the ScanWatch and the Sleep Mat. The ScanWatch provides activity and cardiovascular data, including step counts and heart rate readings, each associated with their corresponding timestamps. The Sleep Mat captures detailed sleep-related metrics, including sleep state (with start and end timestamps), as well as sleep physiology parameters such as heart rate, respiration rate, snoring events, and the standard deviation of heart rate calculated over one-minute windows, all timestamped accordingly.
As shown in Fig. 1, users authenticate through the Withings Health Mate application11, granting permission for data access. The authentication process is managed by the processing infrastructure, which generates an authorisation link and redirects users accordingly to the Resilient web application. Once authorisation is granted, the processing unit extracts the authorisation code, enabling secure data acquisition.
RESILIENT platform architecture: an open-source digital solution designed to integrate data from remote healthcare monitoring. The platform integrates data from Withings wearable devices. The processing infrastructure handles secure data acquisition, storage in a relational database, and the generation of healthcare and usage PDF reports. The web-based application provides healthcare professionals with an intuitive interface for data visualisation, report generation, and raw data access.
At the core of the Resilient’s processing infrastructure lies a dedicated module responsible for handling healthcare data. This module processes incoming data streams and stores them in a relational database system, which was developed using the Django web framework. The database is structured to support efficient querying, secure data management, and seamless integration with reporting and monitoring services. The stored data is subsequently used to generate detailed PDF reports containing clinically relevant health metrics. In parallel, the infrastructure incorporates a monitoring component that continuously tracks the status of connected wearable devices, including their most recent readings, connectivity status, and battery levels.
Complementing the processing infrastructure, the Resilient platform features a web-based application specifically designed for healthcare professionals. It serves as the primary user interface, enabling users to manage and monitor devices, generate and download reports, and visualise patient health data through interactive dashboards. Additionally, the application offers direct access to raw data, enabling more advanced clinical analyses. Communication between the processing module and web application is managed via secure APIs, ensuring real-time data synchronisation and system responsiveness.
Beyond its core functionalities, the platform is designed to be both adaptable and open-source, allowing for seamless modifications to accommodate specific device requirements. This flexibility ensures compatibility with other wearable technologies while preserving an intuitive and user-friendly experience. In line with the FAIRness considerations outlined in Table 3, the platform supports interoperability at the feature level (e.g., heart rate, sleep stages), enabling integration with data from other devices. Although the current implementation is based on Withings APIs11, the underlying data format is structured to allow retrospective harmonisation, and can serve as a template for integrating other wearable APIs used by external researchers. By combining secure data acquisition and visualisation to generate weekly or monthly reports for each individual, the platform serves as a robust tool for remote healthcare monitoring.
Participants and ethical approval
Participants eligible for the study are individuals aged 65 years or older with a diagnosis of at least two chronic health conditions that increase dementia risk (e.g., arthritis, chronic kidney disease, chronic obstructive pulmonary disease, heart disease or failure, depression or other mental health disorders, diabetes, hypertension, and liver disease, stroke) who may participate with or without a study partner. Demographic characteristics of the study participants, including age, sex, and top comorbidities, are summarised in Table 4. Recruitment was conducted through clinicians at Frailty Hubs, UK National Health Service (NHS) hospitals, and GP surgeries in the southeast of England. Clinicians referred eligible patients or performed database searches and sent invitation letters via post, email, or text message. Interested individuals contacted the research team to register their intent to join the study. Individuals with severe mental health (e.g., severe depression, psychosis, agitation, or anxiety), severe sensory impairment, treatment for a terminal illness at baseline (with a life expectancy of less than six months or being in the last year of life), or an inability to provide informed consent were excluded from the study. All potential participants received a Participant Information Sheet outlining the nature of the study, data usage and retention policies, and their rights under the General Data Protection Regulation (GDPR). They were given at least 24 hours to consider participation and encouraged to consult with family or caregivers before providing written informed consent. Capacity to consent was assessed in accordance with Good Clinical Practice and the Mental Capacity Act 2005. Consent included agreement to participate and to the collection, storage, and sharing of anonymised and pseudonymised data in compliance with NHS and GDPR data protection standards. The principal investigator reviewed individual cases to determine final eligibility.
The RESILIENT study has been reviewed and approved by the London-Surrey Borders Research Ethics Committee and the Health Research Authority and is registered on the Integrated Research Application System (IRAS) under reference number 321104. This publicly available dataset includes remote healthcare monitoring data and baseline mental health and cognitive assessments conducted throughout the monitoring period, providing a comprehensive resource for analysing health trends and detecting early signs of cognitive and physiological decline.
Dataset Collection
We integrated wearable and in-home sensory data with individuals’ healthcare information extracted from REDCap12,13 records to create a comprehensive view of the participants’ well-being and care needs.
Physiological data was acquired through wearable and sleep mat devices, which continuously collected and transmitted data to the RESILIENT platform. Within the platform’s processing infrastructure, data from each device was preprocessed, de-identified, cleaned (removing redundant records), and merged according to the source device. A detailed explanation of the available data is provided in the Data Records.
Mental health, demographics and cognitive assessments for this study were conducted by a monitoring team that contacted participants directly. The dataset includes baseline assessments for ACE-III, PHQ-9, GDS-15, and GAD-7, with 6-month follow-up data available only for ACE-III. The ACE-III is a cognitive screening tool that assesses five domains: attention, memory, fluency, language, and visuospatial function, aiding in the detection of cognitive impairment and dementia14. The PHQ-9 is a self-report measure used to assess the severity of depressive symptoms based on the Diagnostic and Statistical Manual of Mental Illnesses (DSM-5 criteria)15. The GDS-15 is a short, validated screening tool designed to detect depressive symptoms in older adults16. The GAD-7 is a seven-item self-report questionnaire that evaluates the severity of generalised anxiety symptoms, widely used in clinical and research settings17. Table 5 presents the scoring scales for each assessment.
Dataset de-identification
A two-stage de-identification process was applied to the data. In the first stage, the data was pseudo-anonymised to develop analytical methods for the study. In the second stage, data was fully anonymised by removing all personally identifying information and any identifiable attributes. Participants are randomly assigned a Universally Unique Identifier (UID) to enhance security during de-identification. This ensures demographics and raw monitoring data from sleep mats and scan watches cannot be traced back to individuals while preserving the data’s utility for analysis.
Throughout the project, robust information governance, control methods, and procedures were implemented. An NHS-approved Data Processing and Impact Assessment was conducted to oversee the data collection, storage, and access procedures.