As AI accelerates target discovery and pushes more candidates toward the bench, assay development and transfer have become critical bottlenecks. Teams relying on one-factor-at-a-time (OFAT) workflows struggle to tame biological complexity, and the field’s widely acknowledged reproducibility problem saps time, confidence, and budgets. Against this backdrop, Synthace and Charles River Laboratories (CRL) have announced a collaboration that brings design-of-experiments (DOE), automation, and software into routine assay development and transfer.
Few people are closer to this than Dr. Markus Gershater, CEO and co-founder at Synthace, a biochemist‑turned business leader who has spent his career engaged in software-driven, multivariate experimentation. He argues that “High Dimensional Experimentation (HDE)”—a next-generation DOE approach at screening scale—can compress timelines and improve transferability by exploring many variables systematically and simultaneously.
HDE is a next‑generation approach to experimental design that replaces OFAT methods with structured, multivariate workflows. Instead of varying individual parameters in isolation, HDE enables scientists to explore many experimental factors systematically, simultaneously, and at scale. Powered by DOE mathematics, automation, and software, HDE generates comprehensive datasets that map how factors interact, revealing optimal conditions and failure modes far earlier in development.
By capturing this broader design space in a single, well‑planned run, HDE reduces iteration, strengthens reproducibility, and produces assays that transfer more reliably.
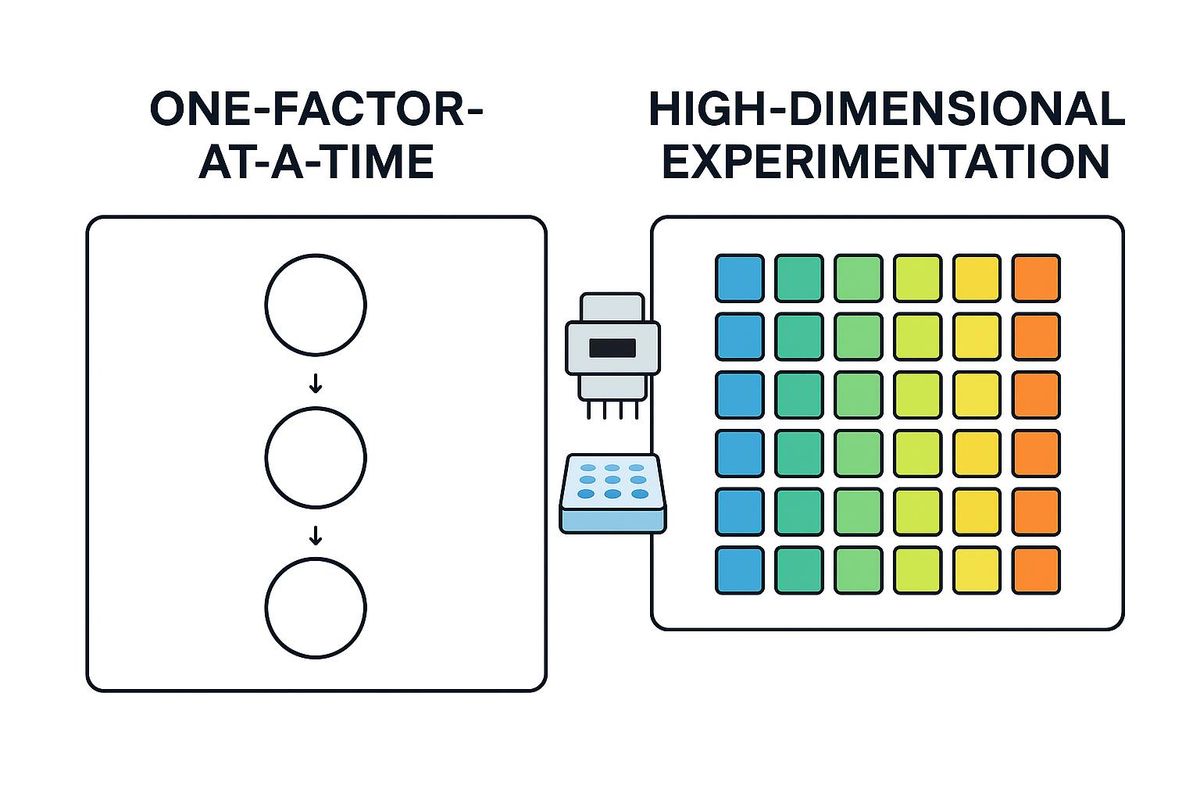
Figure 1. Comparison of OFAT and HDE workflows. Traditional OFAT proceeds step‑by‑step, whereas HDE uses liquid‑handling automation and multivariate designs to screen hundreds of conditions at once. Credit: AI-generated image created using Microsoft Copilot (2026).
In this article, Gershater explores why HDE matters, discusses the results of an HDE proof-of-concept (PoC) study, and explains how digitized, automation-ready designs produce assays that are faster to build and easier to transfer—with clearer traceability for audits and scale-up.
Why the Synthace and Charles River collaboration is timely for drug discovery
Synthace’s collaboration with Charles River gives CRL’s discovery clients access to Synthace’s approach for developing, automating, and transferring assays. In a PoC, CRL used Synthace to develop a 1536-well assay that explored >700 conditions. This method was then transferred across different instruments and sites with comparable results. For a domain in which 72% of researchers acknowledge a reproducibility crisis, Gershater frames this as a way to reduce costly iterations and delays.
“We’re bringing high-dimensional experiments to hard problems like assay development so teams can test a huge number of conditions in parallel, cut timelines, and really raise quality.” — Dr. Markus Gershater
What the Synthace and CRL collaboration means for labs:
- The collaboration operationalizes DOE, automation, and software for CRL programs.
- The automation-ready design could shorten development cycles and improve confidence in transfer.
From OFAT to HDE: What changes in practice?
Most scientists learned to vary OFAT to infer causality. HDE retains rigor but varies multiple factors simultaneously in a structured design, then deconvolutes effects with appropriate analysis—made possible by miniaturization and automation.
“It’s a well-established branch of mathematics… what makes this special is miniaturizing and automating it so you can run designs with hundreds of unique conditions you’d never try by hand.” — Dr. Markus Gershater
In the CRL PoC, a single run in a 1536-well format explored ~seven factors over >700 unique conditions. Instead of weeks of serial iteration, the team obtained an explicit map of the design space, not just a “best” recipe—knowledge that later smoothed assay transfer.
How HDE improves assay development outcomes:
- Captures interactions OFAT typically misses, improving robustness.
- Software and automation make large designs run‑ and audit‑ready.
- Delivers design‑space understanding that underpins reliable transfer.
Compressing assay development timelines
Gershater notes that very complex assays can take up to nine months to develop with conventional methods; HDE has knocked up to six months off in some programs. For simpler biochemical assays, development can often be collapsed into a single experiment—on the order of days to two weeks—because teams explore the space upfront rather than hoping early choices are lucky.
“If there is a way to get the assay up and running, you’ll find it immediately when you test the whole design space upfront.” — Dr. Markus Gershater
HDE can have the following impacts on assay development timelines:
- Complex assays: up to six months saved in some cases.
- Simpler assays: single experiment development in 384/1536 formats.
- Parallel exploration reduces “got lucky” risk and rework loops.
Replicating results across sites, instruments, and people
Assay transfer often fails on tacit steps omitted from standard operating procedures. In the PoC, CRL developed a 1536-well assay in one lab, then reproduced results on a different instrument make and model, with different personnel and reagent lots—while also discovering that the conventional recipe used previously contained unnecessary reagents that HDE identified as noncontributory.
Gershater’s diagnosis is simple: transfer issues often reflect gaps in understanding. Design space maps, plus a granular digital record of every step, close those gaps.
Gershater ties reproducibility failures to the loss of experimental context in the form of undocumented, informal choices that shape outcomes. In Synthace’s workflow, scientists declare factors; the platform designs the experiment, drives automation, and logs every liquid transfer to every well from every stock. Each result is linked to its complete provenance, which becomes critical for troubleshooting, method transfer, and audits.
“You have a fantastically detailed experimental map… for every one of the 1536 wells, you know exactly how that data point was produced.” — Dr. Markus Gershater
Where HDE goes next: complex biology, 3D models, and scale‑up
While HDE “blasts through” simpler assays, Gershater expects the greatest gains to be in cell-based assays and increasingly 3D systems and organoids, where multiparameter interactions defeat OFAT. Charles River plans to standardize HDE across discovery functions, so clients see faster, higher-quality outcomes program-wide, and to connect wet lab designs to AI-driven loops as validation becomes the rate-limiting step.
Gershater’s final reflection provides a lens through which to view the potential broader impact of HDE on modern discovery workflows.
“Biology demands multivariate thinking… my personal hope and ambition is to see this technology enabling progress with ever more sophisticated in vitro systems. That’s where we have an opportunity to lift the ceiling.” — Dr. Markus Gershater
Key takeaways:
- HDE (DOE, automation, and software) compresses timelines and improves robustness vs OFAT.
- The CRL PoC demonstrated the design of a 1536‑well assay, capable of exploring >700 conditions that transferred across sites/instruments with comparable results.
- Digitized provenance per well strengthens reproducibility, transfer, and audit readiness—directly addressing a widely recognized pain point in drug discovery.
This content includes text that has been created with the assistance of generative AI and has undergone editorial review before publishing. Technology Networks’ AI policy can be found here.