NTM-KOREA is an ongoing nationwide prospective cohort study that began on February 28, 2022 (ClinicalTrials.gov identifier: NCT03934034)34. Patients starting treatment for NTM-PD were enrolled from eight institutions across South Korea. The eligibility criteria were: (1) aged ≥ 19 years; (2) diagnosed with NTM-PD per current guidelines4; (3) having one of the following etiologic organisms: Mycobacterium avium complex, Mycobacterium abscessus (subspecies abscessus, sub. massiliense), or Mycobacterium kansasii; and 4) ≤ 4 weeks of starting antibiotic treatment.
The study protocol was approved by the institutional review boards of all participating institutions prior to participant enrollment:
-
Seoul National University Hospital (IRB No. H-1903-021-1015, approved on March 19, 2019).
-
Pusan National University Hospital (IRB No. 1911-005-084, approved on November 14, 2019).
-
Samsung Medical Center (IRB No. SMC 2019-06-030, approved on June 13, 2019).
-
Asan Medical Center (IRB No. 2019 − 0638, approved on May 9, 2019).
-
Severance Hospital, Yonsei University College of Medicine (IRB No. 4-2019-0297, approved on May 20, 2019).
-
Pusan National University Yangsan Hospital (IRB No. 05-2019-081, approved on May 22, 2019).
-
Chonnam National University Hospital (IRB No. CNUH-2019-179, approved on June 25, 2019).
-
Seoul National University Bundang Hospital (IRB No. B-2111-722-402, approved on November 3, 2021).
All participants provided written informed consent, and all procedures were conducted in accordance with the approved protocol, the relevant guidelines and regulations, and the ethical principles of the Declaration of Helsinki. Participants in the NTM-KOREA cohort were included in our previous analysis33.
Data collection
This study was a cross-sectional analysis based on the baseline data from the NTM-KOREA cohort. All variables, including the BACES score, HRQOL, nutritional and physical assessments, as well as functional outcomes, were collected at the time of cohort enrollment, which was within four weeks of antibiotic treatment initiation. We gathered demographic and clinical data during enrolment from the registered databases of NTM-KOREA (iCreaT ver. 2.0 [https://www.icreat/nih.go.kr]). Baseline characteristics included age, sex, BMI, smoking history, and comorbidities. Microbiological data included etiological organisms and smear results. Local thoracic radiologists or experienced pulmonary physicians interpreted chest computed tomography scans and classified them as the nodular bronchiectatic or fibrocavitary type. Radiologic subtypes not belonging to these types, such as focal cavity, nodule, mass, or consolidation, were designated as unclassifiable35.
BACES score
The BACES score was calculated with BMI < 18.5 kg/m2, age ≥ 65 years, presence of cavities, elevated erythrocyte sedimentation rate, and male sex each equating to 1 point11. Patients with a score of 0–1 were classified into the mild group; 2–3, moderate group; and 4–5, severe group12,13,14.
HRQOL and associated factor measurement
HRQOL is associated with various clinical variables, including nutritional status, sarcopenia, and physical activity29,31,36. The following data were collected to measure HRQOL and associated health conditions: (1) respiratory or constitutional symptoms; (2) self-reported data using the QOL-B questionnaire, MNA-SF questionnaire, and IPAQ-SF; (3) laboratory-based data of PNI; and (4) measurement-based data, such as 6MWD, handgrip strength, body composition, and spirometry measurements (FEV1, FVC, FEV1/FVC, and DLCO).
The HRQOL of patients with NTM-PD was measured using the QOL-B questionnaire, which comprises 37 items covering respiratory symptoms, physical functioning, vitality, role functioning, health perception, emotional functioning, social functioning, and treatment burden16. The score range for each domain is 0–100, with higher values indicating a better quality of life. The Korean-translated version of the QOL-B questionnaire was used with permission from Dr. Alexandra L. Quittner. The questionnaire was administered in paper form under the supervision of trained nurses, as previously described33.
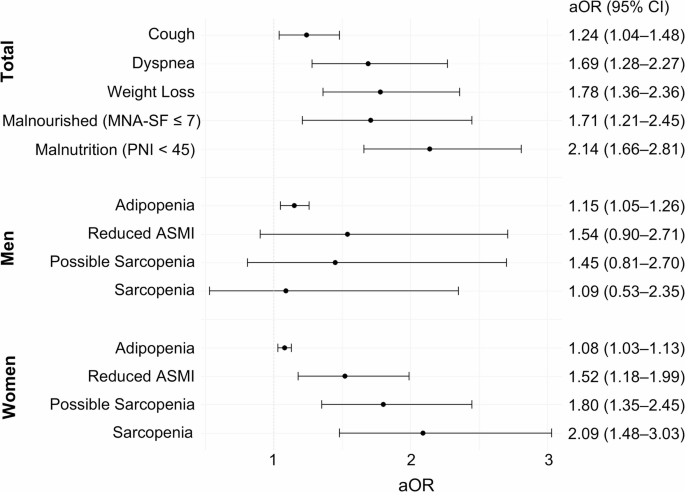
Nutritional status was assessed using MNA-SF and PNI. The MNA questionnaire evaluates dietary intake, recent weight change, neuropsychological issues, and functional abilities37. It is a simple, effective questionnaire for assessing the nutritional status of older adults and patients with respiratory diseases38,39. MNA-SF can effectively identify malnutrition in patients with NTM-PD40. Participants with MNA-SF scores of 12–14 were classified as having a normal nutritional status; 8–11, as being at risk of malnutrition; and ≤ 7, as being malnourished40. The PNI was initially developed for the composite assessment of perioperative nutritional status in patients with malignancies, with its use later extended to various diseases41,42,43. The PNI score is calculated using the following formula: 10 × serum albumin value (g/dL) + 0.005 × total lymphocyte count in the peripheral blood (/mm3). Participants with PNI < 45 were assigned to the malnutrition group44.
Anthropometric data to identify participants with sarcopenia were collected using multi-frequency bioelectrical impedance analysis. Bioelectrical impedance analysis can produce reliable body composition estimates compatible with those measured using dual-energy X-ray absorptiometry45. The detailed models used are presented in Supplementary Table S1. Adipopenia is defined as a fat mass index (total body fat mass divided by the square of height) < 3.0 kg/m2 for men and 5.0 kg/m2 for women19. Handgrip was measured using a Jamar® hydraulic hand dynamometer (Model 5030J1; Sammons Preston, Bolingbrook, IL, USA). Possible sarcopenia was defined as reduced handgrip strength (< 28 kg for men and < 18 kg for women) according to the Asian Working Group for Sarcopenia (2019)20. Sarcopenia was diagnosed if patients also had reduced ASMI (sum of skeletal muscle mass in the arms and legs divided by the square of height: <7.0 kg/m2 for men and 5.7 kg/m2 for women)20.
Physical activity was evaluated with the IPAQ-SF questionnaire that collects information on the time spent in sedentary activities, walking, as well as in moderate- and vigorous-intensity activities over the past 7 days. IPAQ-SF has demonstrated feasibility in national and regional prevalence studies46.
All questionnaires and assessment tools used in this study were either used with appropriate permissions (QOL-B) or were publicly available for research purposes (MNA-SF, IPAQ-SF) and were implemented in accordance with their respective usage guidelines.
Statistical analysis
Data are presented as median with interquartile range for continuous variables and as proportion for categorical variables. The Kruskal–Wallis test was performed to compare continuous variables among multiple groups. Post-hoc comparisons were performed using Dunn’s test. To control for the increased risk of type I errors due to multiple comparisons, the Bonferroni correction was applied. Fisher’s exact test was performed to compare categorical variables. For the BACES mild, moderate, and severe groups, the Cochran–Armitage trend test was performed to assess trends across the ordered groups. Linear and logistic regression analyses were performed to identify factors associated with BACES scores as well as with HRQOL and related factors. Clinically relevant variables based on previous studies were included as covariates in the models (e.g., smoking status, tuberculosis history, respiratory comorbidities, MNA-SF, and PNI). Given that the BACES score already includes age, BMI, and sex as components, these variables were not included separately in the multivariable models to avoid collinearity. P < 0.05 indicated statistical significance. All statistical analyses were conducted using the R software (v.4.3.1; The R Foundation for Statistical Computing, Vienna, Austria).