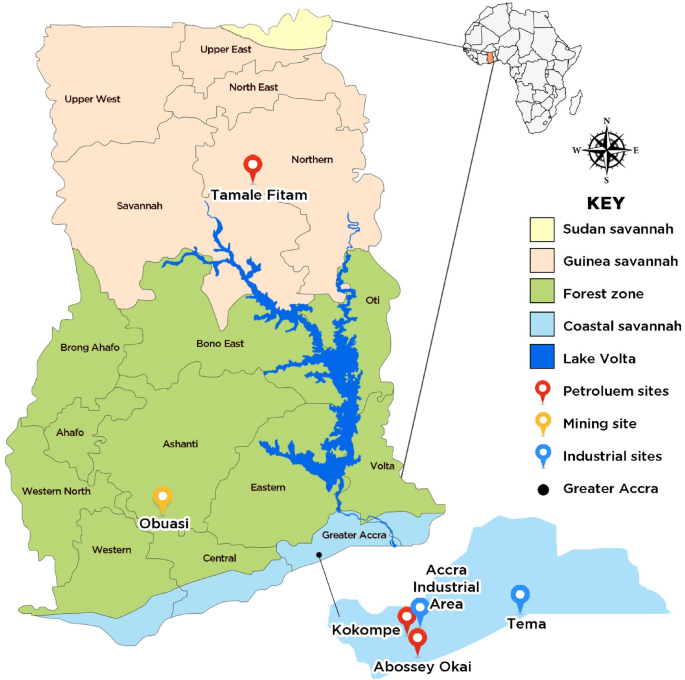
This study was conducted across six sites in Ghana, selected to represent diverse ecological zones and environmental stressors that could facilitate the mediation of insecticide resistance in Anopheles mosquitoes (Fig. 3). These sites were categorized into informal vehicle repairing sites where petroleum gets spilt through the repair of cars (herein after: petroleum sites category) which were: Abossey Okai, Kokompe, Tamale Fitam; sites where surface mining is undertaken (mining site category: Obuasi) and sites with industries where effluents are spilt in the environment (Industrial sites category: Tema community 1, Accra Industrial Area).
Map of Ghana showing the various study sites (The base map for the study site depiction was sourced from https://ghana-mission.co.in/mapofghana/ and modified using Adobe Photoshop (Version 7.0.1)).
Abossey Okai (5.5480° N, 0.2424° W) and Kokompe (5.5813° N, 0.2173° W) are both located in the city of Accra, in the coastal savannah zone of Southern Ghana. Oil spills as a result of engine oil changes and vehicle repairs, and the leaching of metal compounds such as lead (Pb), cadmium (Cd), and chromium (Cr) from sources like lead pipes, used batteries, brake pads, and metal scraps commonly found in vehicle repair garages into mosquito breeding habitats at these sites may trigger adaptive responses and contribute to increased insecticide resistance in the vectors. Tamale Fitam (9.4198° N, 0.8199° W), located in the Guinea savannah zone of Northern Ghana, may also have oil spills as a result of automobile repair activities similar to Abossey Okai and Kokompe.
Obuasi (6.2024° N, 1.6658° W), located in Ghana’s middle forest zone, is known for its intensive deep and surface mining for decades. These mining operations create pockets of breeding habitats for Anopheles mosquitoes. Processing the gold ore with mercury, zinc, cyanide and other heavy metals introduces toxic contaminants into the environment, which may leach into larval habitats and exert chronic selection pressure on mosquito populations. Moreover, there has been a prolonged IRS campaign by the AngloGold Ghana malaria control program, spanning nearly two decades (2006–2024), during which mosquito populations in Obuasi have been exposed to successions of different insecticide classes. These include organophosphates and carbamates (e.g., pirimiphos-methyl, extensively used from 2006 to 2017), pyrethroids, neonicotinoids, neonicotinoid + pyrethroid combinations (2018–2023), and more recently, meta-diamides. The sequential and overlapping intense selection pressures from both environmental contaminants and long-term insecticide use may be key mediators of insecticide resistance in Anopheles gambiae s.l.
Tema Community 1 (5.6698° N, 0.0200° W), located in the coastal savannah of southern Ghana, is an industrial and port city. The dense concentration of manufacturing facilities and port operations generates significant industrial effluents, many of which are discharged into open drains and stagnant pools that double as mosquito breeding grounds. Similarly, Accra Industrial Area (5°33’16.992’’ N, 0°13’15.492’’ W), located in the city of Accra, is a densely populated industrial zone characterized by a high concentration of manufacturing facilities. These factories are potential sources of atmospheric pollutants and chemical effluents, which often discharge into open drains and stagnant water bodies. the presence of these pollutants in drainages where mosquitoes could be breeding may exert selection pressures capable of inducing or enhancing insecticide resistance in local Anopheles vector populations.
The coastal savannah in southern Ghana has a tropical savannah climate with temperatures ranging from 23 to 34 °C and an average annual rainfall of 787 mm, following a bimodal pattern with rainy seasons from April–June and October–November. The dry season lasts from December to March. The forest zone of Ghana experiences a tropical forest climate with 1,500–2,000 mm of annual rainfall, also in a bimodal pattern, with rainy seasons from March–July and September–November, and dry periods from August–February. Temperatures remain stable between 24 and 30 °C. The Guinea savannah in the north of Ghana has a unimodal rainfall pattern from May–November, averaging 900 mm annually. The dry season (December–April) sees temperatures rising to 42 °C, with a mean annual temperature of 28 °C.
Mosquito larval collection and raising in the insectary
Anopheles larvae sampling was carried out from January 2023 to July 2024. To avoid the collection of sibling species, larvae were sampled randomly from different breeding habitats in each study site. This study was approved by the Ethics and Protocol Review Committee of the College of Health Sciences, University of Ghana (protocol identification number: CHS-Et/M.8-P4.6/2023–2024). All methods were carried out in accordance with relevant guidelines and regulations. Meetings were held at each study site with chiefs, community leaders, and residents to introduce the research. Permission to conduct the study at the various sites was obtained from community leaders. Verbal informed consent was obtained from community leaders and residents for mosquito sampling activities. Immature Anopheles mosquitoes collected from the same site within the Sahel-savannah zone were transported to the insectary of the President’s Malaria Initiative (PMI) Project Office in Tamale. Similarly, larvae collected from sites within the forest and coastal savannah ecozones were transported to the insectary of the AngloGold Ashanti malaria control programme (AGAmal) in Obuasi, and the Department of Medical Microbiology, University of Ghana Medical School, Korle-Bu, Accra, respectively. The mosquito larvae were raised to adults in the insectary, maintained at an average temperature of 28 ± 1 °C and a relative humidity of 80.9 ± 6.3%. Once emerged, the adult mosquitoes were provided with a 10% sucrose solution for feeding. Three to five (3–5) days old sugar-fed, but not blood-fed, females were later selected for WHO susceptibility tests and intensity bioassays.
Phenotypic Resistance in Anopheles mosquitoes using WHO susceptibility tube bioassay
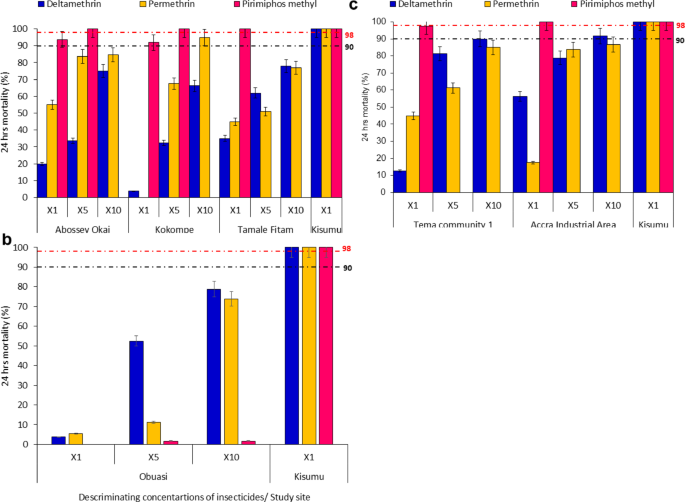
To assess the resistance intensity of An. gambiae s.l. population across various site categories, a batch of 25 non-blood-fed females that were 3–5 days old were subjected to the WHO susceptibility test bioassay. Four replicates and 2 controls were used for each insecticide using the standard WHO tube assay procedure38,39. Mosquitoes were morphologically identified as belonging to the An. gambiae s.l. complex. The WHO test papers impregnated with insecticides at discriminating concentrations used were obtained from the WHO Collaborating Centre at Universiti Sains Malaysia. These included: permethrin [1 × (0.75%), 5 × (3.75%), and 10 × (7.5%)], deltamethrin [1 × (0.05%), 5 × (0.25%), and 10 × (0.5%)], and pirimiphos-methyl [1 × (0.25%), 5 × (1.25%), and 10 × (2.5%)]39. The insecticide papers were initially tested on the Kisumu susceptible strain, a reference strain susceptible to insecticides, to confirm their efficacy.
Phenotypic resistance to chlorfenapyr and clothianidin in Anopheles gambiae s.l. using WHO susceptibility bottle bioassay
To determine the susceptibility status of Anopheles gambiae s.l. to chlorfenapyr and clothianidin, the WHO Bottle bioassays were carried out using four replicates according to WHO guidelines40; each replicate consisted of 25 female Anopheles mosquitoes aged 3–5 days. These mosquitoes were exposed to 100 µg/ml chlorfenapyr-acetone solution and 4 µg/ml clothianidin-acetone-MERO solution for 1 h; this was done approximately 24 h after coating the bottles with the respective insecticides. Two additional replicates of 25 mosquitoes each served as a negative control, with bottles treated with 1 ml of acetone for chlorfenapyr, or acetone with MERO for clothianidin. The Knocked-down mosquitoes were recorded at the end of the 60 min exposure period. After exposure, mosquitoes were transferred to a paper cup covered with untreated netting and provided with a 10% sugar solution soaked in a wad of cotton, which was changed daily; the assay was monitored and mortalities were recorded at 24 h, 48 h, and 72 h. The Kisumu susceptible strain, maintained under the same insectary conditions, was utilized as the primary test control to monitor the quality of the coated bottles.
Collection, processing and screening for heavy metals and insecticide residues in water samples from Anopheles breeding habitats
Water samples were collected from mosquito breeding habitats located within three environmentally distinct site categories: mining, industrial, and petroleum-contaminated areas. At each site, 50 mL of surface water was collected in sterile Wheaton bottles. Samples were collected in triplicate, stored immediately on ice, and transported to the Ghana Standards Authority (GSA) laboratory in Accra for pesticide and insecticide residue analysis.
In the laboratory, the samples were first filtered to remove debris and suspended solids. Residue extraction was performed using liquid-liquid extraction (LLE) with suitable organic solvents. The extracts were further cleaned using solid-phase extraction (SPE) cartridges to reduce matrix interferences. Following extraction, the samples were concentrated under a gentle nitrogen stream and reconstituted in an appropriate solvent compatible with the analytical system. Residue analysis was conducted using gas chromatography–mass spectrometry (GC-MS) based on validated multi-residue analytical protocols. Chromatographic separation was achieved using a VF-5ms capillary column (30 m × 0.25 mm ID × 0.25 μm film thickness), with an optimized oven temperature program for effective compound separation. The mass spectrometer operated in selected ion monitoring (SIM) mode to enhance detection sensitivity and selectivity. Identification of analytes was based on retention times and mass spectral comparison with certified reference standards and the EPA spectral libraries41. The method achieved a limit of detection (LOD) of 0.01 mg/L and a limit of quantification (LOQ) of 0.05 mg/L, with only values above the LOQ considered for quantification. To ensure the reliability and validity of the analytical results, rigorous quality assurance and quality control measures were implemented. Method, solvent, and procedural blanks were included in each analytical batch to monitor contamination. Matrix spike recoveries were used to assess extraction efficiency, with acceptable recovery rates ranging from 70 to 120%. Certified reference materials (CRMs) and internal standards were employed to verify accuracy and precision, while calibration curves prepared with at least five concentrations of analytical standards demonstrated strong linearity (R² ≥ 0.995). Replicate analyses were also performed to ensure reproducibility.
In addition to the insecticide residue analysis, the same water samples were screened for heavy metals. Samples were immediately acidified to pH < 2.0 with 1 mL of concentrated HNO₃ to preserve dissolved metals and inhibit microbial activity. In the laboratory, acid digestion was performed to prepare the samples for total recoverable metal analysis. Specifically, 100 mL aliquots of each sample were mixed with 50 mL of concentrated nitric acid (1:1 v/v) and gently heated to reduce the volume to approximately one-third. After cooling, 20 mL of hydrochloric acid (1:1 v/v) was added, followed by a second digestion step at low heat for 10 min. The digests were filtered, diluted to 100 mL with deionized water, and stored in clean glass containers. Quantitative analysis of heavy metals, including cadmium (Cd), chromium (Cr), copper (Cu), iron (Fe), lead (Pb), manganese (Mn), and zinc (Zn), was conducted using a Perkin Elmer NexION 2000 Inductively Coupled Plasma Mass Spectrometer (ICP-MS), equipped with a micro-mist nebulizer, quartz spray chamber, and Peltier cooler. The analysis followed the U.S. EPA Method 200.8 for total recoverable metals41. The instrument was calibrated with certified multi-element standards, and quality control was ensured through the inclusion of method blanks, matrix spikes, and duplicate samples41.
Detection of target-site mutations in An. gambiae s.l.
Conventional and real-time PCR were used to investigate the presence of insecticide resistance genes, including kdr haplotypes (L995F, L995S, N1570Y, V402L, I1527T, and P1874L) and Ace-1R G280S42. The allele-specific PCR procedure for kdr genotyping was used to detect kdr alleles using the protocol and primer sequences described by Martines-Torres et al.. and Jones et al.43,44. for N1570Y; Williams et al.45. for V402L, I1527T and P1874L; and Weil et al.46. for the Ace-1R G280S mutation.
Characterization of Anopheles gambiae s.l.
A sub-sample of the mosquitoes after the phenotypic susceptibility testing were randomly selected and identified morphologically using the keys of Gillies and Coetzee47. Members of the An. gambiae s.l. were further identified by PCR to distinguish sibling species using a leg of each individual mosquitoes, as previously described by Scott et al.48. and PCR-RFLP by Fanello et al.49.
Data management and analysis
Descriptive analysis was done to visualize WHO susceptibility data, resistant allele frequencies, and mosquito species composition from the selected sites using graphs and tables.
WHO insecticide susceptibility levels were classified using the WHO criteria40,50. Allele frequencies of resistance gene markers in the vector populations at each site were calculated using Hardy-Weinberg equilibrium (HWE), with the formula F (allele frequency) = (2nRR + nRS) / 2 N. Logistic regression was used to assess the association between insecticide resistance with categorical data and the presence of heavy metals. The concentration and identification of pesticide residue compounds and heavy metals were determined using the U.S. EPA Method 200.8 for total recoverable metals reference standards41relative retention times and mass-to-charge ratios (GS-MS, model QP2010). P ≤ 0.05 was considered significant. All statistical analyses were done in R 4.2.2 via RStudio (2022.12.0 + 353) and STATA/IC 14.1.