image:
- Offers a thorough review on the mechanism of molecular and ion dipoles in high-energy batteries, covering development, classification, and multifaceted roles in battery systems.
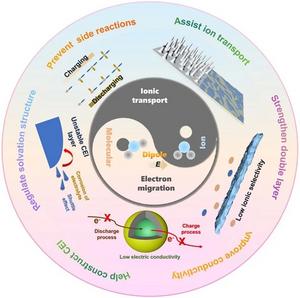
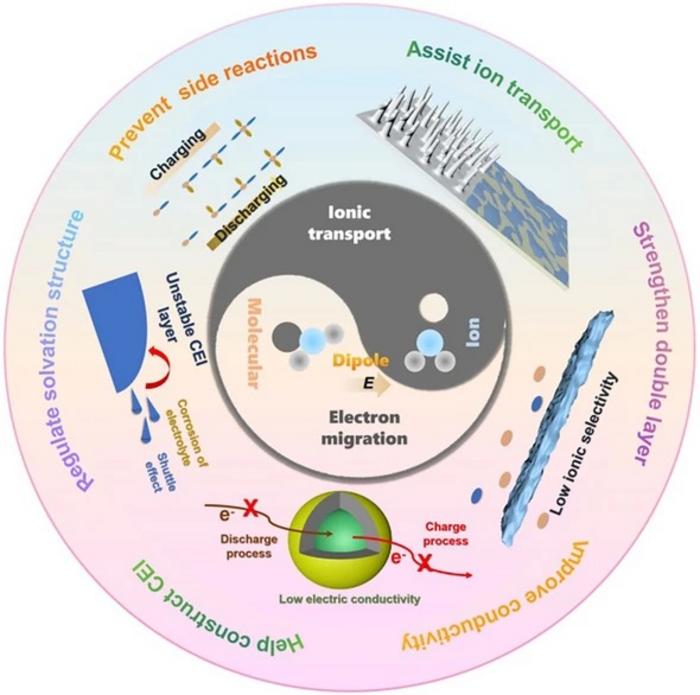
- Elucidates how molecular and ion dipoles regulate ionic transport, optimize solvation structures, strengthen the electric double layer, and construct stable solid electrolyte interphase/cathode–electrolyte interface layers, all of which boost battery performance.
- Demonstrates the wide-ranging applications of dipole interactions in various battery systems, such as suppressing dendrites in lithium–metal batteries and improving the cycling stability of lithium–sulfur batteries.
- Proposes future research directions including AI-assisted materials design, in-depth mechanism exploration, multidisciplinary integration, database establishment, and promoting practical applications, aiming to drive the development of high-energy batteries.
Credit: Shihai Cao, Yuntong Sun*, Yinghao Li, Ao Wang, Wenyao Zhang, Zhendong Hao*, Jong-Min Lee*.
As global demand for sustainable energy surges, the performance ceiling of current battery technologies is increasingly tied to how efficiently ions and electrons move through the cell. Now, a multinational team led by Dr. Yuntong Sun (Nanyang Technological University), Dr. Zhendong Hao (Nanjing Institute of Technology) and Prof. Jong-Min Lee (DGIST) has delivered a panoramic review in Nano-Micro Letters showing how molecular and ionic dipole interactions can push that ceiling higher. The work provides a design playbook for next-generation high-energy batteries that are safer, longer-lasting and wide-temperature-capable.
Why Dipole Interactions Matter
- Energy Density Unlocked: Dipole fields regulate ion-solvent coordination, suppress dendrites, stabilize electrode–electrolyte interfaces and unlock extra capacity from existing cathode chemistries.
- Interface Engineering: Dipoles build robust solid-electrolyte interphase (SEI) and cathode–electrolyte interphase (CEI) layers, cutting parasitic reactions and impedance growth.
- Universal Tool-box: From Li-ion, Li-metal and Li–S to Na-ion and Zn systems, dipole strategies display chemistry-agnostic adaptability across liquid, gel and solid-state formats.
Innovative Design and Features
- Dipole Classifications: Ion–solvent molecule, ion–functional group and additive molecule ion–dipole interactions are dissected with structure–function tables linking specific dipole motifs to performance gains.
- Functional Materials: Crown ethers, ferroelectric BaTiO3, polar carbonates, sulfonamides and nitrile-rich polymers are spotlighted as dipole donors that re-wire solvation sheaths and electric-double-layer topology.
- Array Architectures: Electric-field-assisted vertical alignment, in-situ UV polymerization and asymmetric ceramic/polymer integration create oriented ion highways inside composite electrolytes and separators.
Applications and Future Outlook
- Multi-Level Transport: Dipole-ordered channels raise Li⁺/Na⁺/Zn2⁺ transference numbers (up to 0.82), cut desolvation barriers and enable 5C–10C fast charge without dendrite initiation.
- High-Voltage Stability: Dipole-engineered CEI layers deliver 91 % capacity retention after 100 cycles at 4.3 V (Li||NCM523) and extend oxidative stability of polymer electrolytes to 4.6 V.
- Wide-Temperature Resilience: Strong multiple ion–dipole networks preserve solvation geometry from −60 °C to 100 °C, yielding 89 % capacity retention at 100 °C and 76 % at −40 °C.
- Challenges and Opportunities: The review flags needs for AI-aided dipole design, in-situ characterization databases and scale-up collaboration to translate dipole-boosted coin-cell records into pouch-cell products.
This roadmap underscores the pivotal role of dipole interactions in bridging materials science, electrochemistry and computation for future high-energy storage. Stay tuned for more field-advancing work from Prof. Sun, Prof. Hao and Prof. Lee’s teams!
Nano-Micro Letters
News article
Multifunctional Dipoles Enabling Enhanced Ionic and Electronic Transport for High‑Energy Batteries
5-Jan-2026
Disclaimer: AAAS and EurekAlert! are not responsible for the accuracy of news releases posted to EurekAlert! by contributing institutions or for the use of any information through the EurekAlert system.