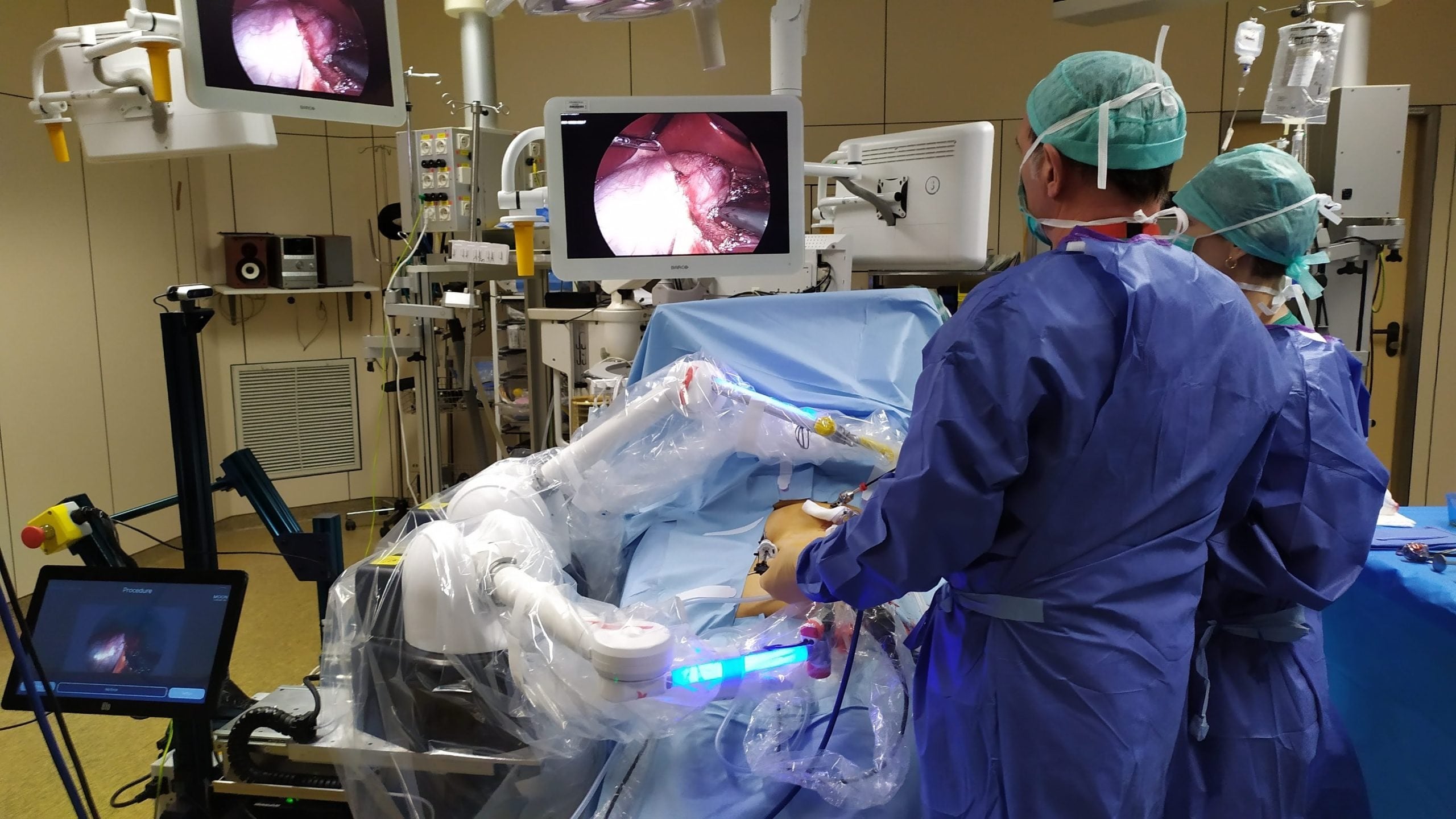
FDA grants clearance for commercial version of Moon Surgical’s Maestro robot – The broad laparoscopy market surgical system supports surgeons in soft tissue surgical procedures not currently supported by available telerobotic systems.
https://www.medicaldevice-network.com/news/fda-grants-clearance-for-commercial-version-of-moon-surgical-maestro-robot/

1 Comment
From the article
>Moon Surgical has received clearance from the US Food and Drug Administration (FDA) for the commercial version of its Maestro surgical system.
>The company previously received FDA clearance for a non-commercial version of the system in September 2022. In September 2023, Maestro was used to assist in [weight loss surgery procedures in the US](https://www.medicaldevice-network.com/news/moon-surgicals-maestro-system-used-in-first-us-clinical-procedures/).
>In addition to its surgical assistance capabilities, Maestro is equipped with the edge computing platform [NVIDIA](https://www.globaldata.com/store/report/?cdmsid=1712947&scalar=true&utm_source=Company%20News&utm_medium=23-188355&utm_campaign=company-profile-hyperlink-nonlgp) Holoscan, which enables real-time algorithms based on AI to be deployed in the operating room for immediate benefits during surgery.